 The CO 2 molecule has a linear geometry, while the H 2 O molecule is said to be bent, forming a broad V-shape with the oxygen atom at its vertex. Carbon dioxide (CO 2 ) and water (H 2 O) are both molecular substances, each of which has a molecular structure consisting of a single atom of one element covalently bonded to two atoms of another element, but they have different geometries. The situation changes when a molecule consists of more than two atoms. And yet the hydrogen fluoride also has a linear geometry, because there is simply no other way to arrange two atoms. A hydrogen fluoride (HF) molecule, on the other hand, has a strong dipole moment because fluorine - the most electronegative element in the periodic table - has an electronegativity of 3.98 compared with a value of 2.20 for hydrogen. As a result, the O 2 molecule has zero dipole moment. The diatomic oxygen (O 2 ) molecule is non polar because the two oxygen atoms have the same electronegativity, and there are no partial charges on either atom. Your answers should be in terms of principles of molecular structure.A simple diatomic molecule consisting of two atoms of the same element will always have a linear geometry. 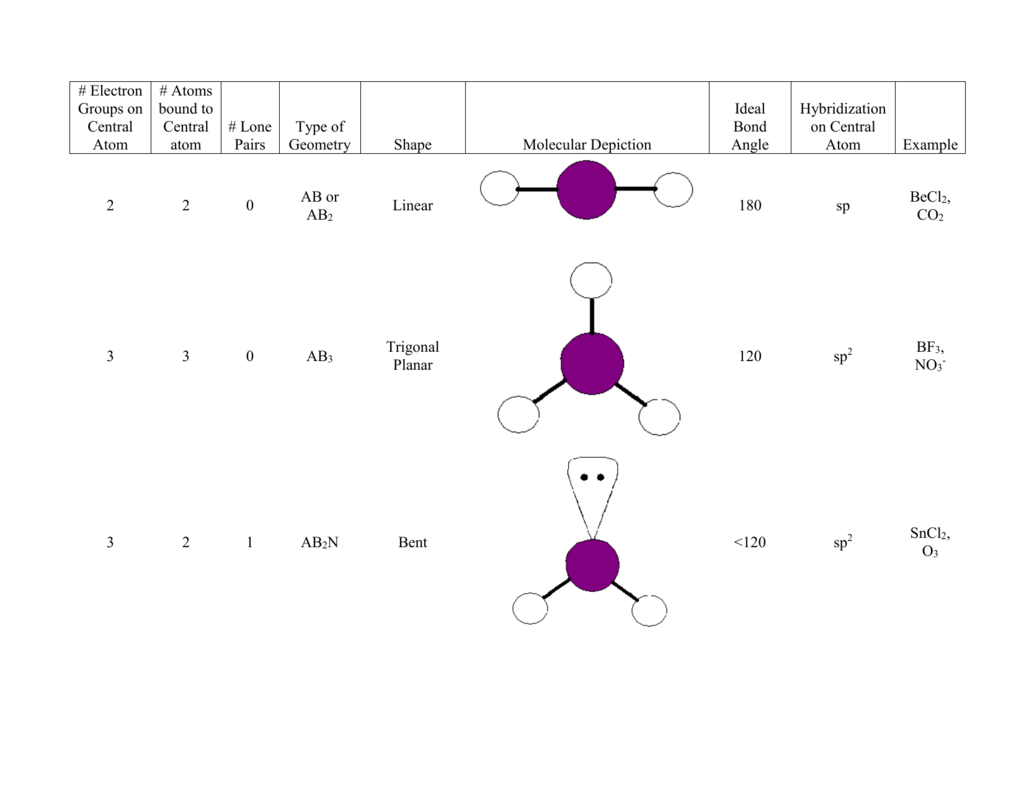
Use the information in the table below to respond to the statements and questions that follow. (d) On the basis of your Lewis electron-dot diagram(s) in part (c), identify the hybridization of the sulfur atom in the SO2 molecule. Explain this observation, supporting your explanation by drawing in the box below a Lewis electron-dot diagram (or diagrams) for the SO2 molecule. (c) In the SO2 molecule, both of the bonds between sulfur and oxygen have the same length. (b) On the basis of the Lewis electron-dot diagram that you drew in part (a), predict the molecular geometry of the IF3 molecule. (a) In the box provided, draw a complete Lewis electron-dot diagram for the IF3 molecule. The following questions are from past AP Chemistry exams that were posted online by College Board. The same principle applies to the other hybridizations. In the above example, carbon's 2p and 2s orbitals fuse into 4 half-filled sp3 orbitals that can make 4 sp3-orbital sigma bonds. Hybridization - You only have to memorize the hybridization of families 2, 3, and 4□. Shape - This is the main column that you should memorize and learn to associate with the general formula, electron domain geography, and hybridization. The graph below also includes angle measures that you should be aware of in that specific molecular geometry. It shows where the electrons or atoms are in relation to the middle atom, M. General Formula - made up of three parts:Įlectron Domain Geography - gives you an idea of what the molecule looks like. Once you practice, the questions that involve the VSEPR Theory become free points□!įamily - think of family as how many groups of atoms or molecules branch off the middle atom (number of x + number of e in the general formula). It gives you everything you need to know about VSEPR and will answer a lot of questions that require memorization on the AP. You should definitely memorize the table below for the AP Exam. It specifically uses the Coulombic repulsion between electrons as a basis for predicting electron arrangement. 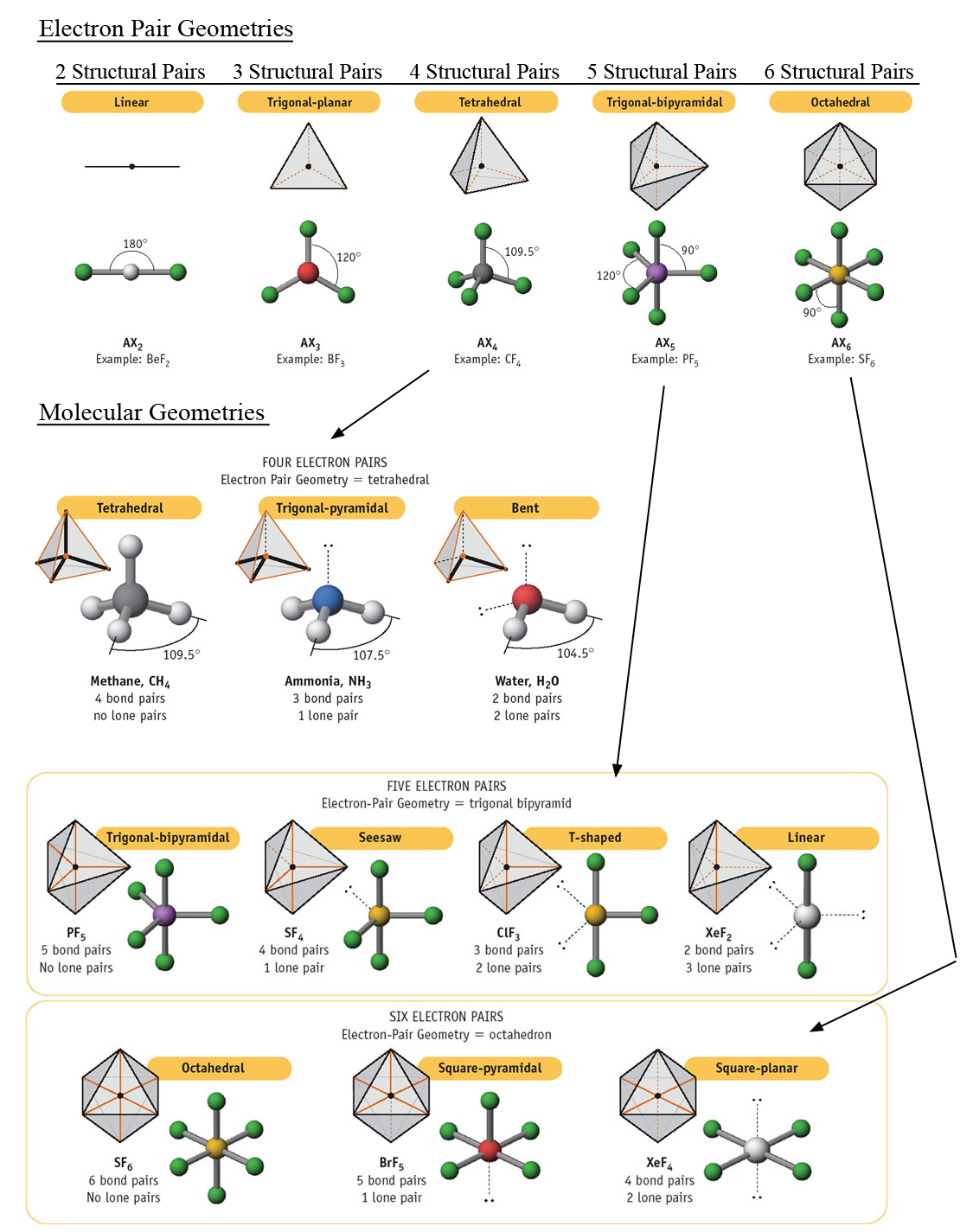
The Valence-Shell-Electron-Pair-Repulsion (VSEPR) theory can predict molecular geometry by minimizing electron-electron repulsion. Lewis structures can determine properties such as geometry, bond orders, bond lengths, and dipoles for molecules. Valence Shell Electron Pair Repulsion (VSEPR) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
